Once classified as a Schedule 1 drug along with all other components of the cannabis plant, CBD is now federally legal… but with some stipulations.
The 2018 Farm Bill provided legal status to Hemp, a strain of cannabis with a very low THC content but typically high levels of CBD. While this has certainly opened many doors for companies and consumers alike, there are some strict regulations governing what kind of CBD products are allowed to be sold/purchased.
‘New’ CBD regulations
According to FDA commissioner Scott Gottlieb,
“It’s unlawful under the FD&C Act (Federal Food, Drug, and Cosmetic Act) to introduce food containing added CBD or THC into interstate commerce, or to market CBD or THC products as, or in, dietary supplements, regardless of whether the substances are hemp-derived.”

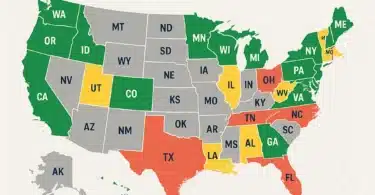
New York has been banning edibles under the ‘new’ CBD regulations
So far, multiple states have banned CBD edibles and supplements under these new regulations: Maine, New York, Ohio, and California to name a few. Even the entire European continent has recently restricted the sale of CBD edibles and supplements, after classifying CBD as a novel food.
Furthermore, it’s also illegal to claim that a specific CBD product can cure or treat any disease or health condition. For a product to be marketed as such, the FDA must first approve it for the intended use.
While it may seem inconvenient, it’s not without good reason. Multiple investigations across the country have determined that quality control for CBD products is questionable at best. On average, 70% of CBD products are mislabeled in some way. Many have less than the amount of THC listed on the label. Others have more THC than what is considered legal, or pesticides, heavy metals, and toxic solvents.
There is also the issue of false or unsubstantiated medical claims. Yes, CBD has many therapeutic benefits, but it’s not the be-all end-all solution to everyone medical problems. A recent review of CBD found that “CBD has been touted for many ailments for which it has not been studied, and in those diseases with evaluable human data, it generally has weak or very weak evidence.”
There are MANY legitimate studies out there, but claiming that CBD can cure a condition for which it hasn’t been researched could be dangerous. And to expand here, there is also the possibility that it could have negative interactions with a patient’s medications that they are already taking. So anyone selling CBD products should tread lightly in this area.
More regulations governing the quality of CBD products will make a huge difference in protecting the people who truly need them for health reasons. For more information about regulations, subscribe to the ‘Medical Cannabis Weekly Report’ our Weekly Newsletter which is full of all the latest news and information about the cannabis industry.




Have anything to add? Your voice matters! Join the conversation and contribute your insights and ideas below.